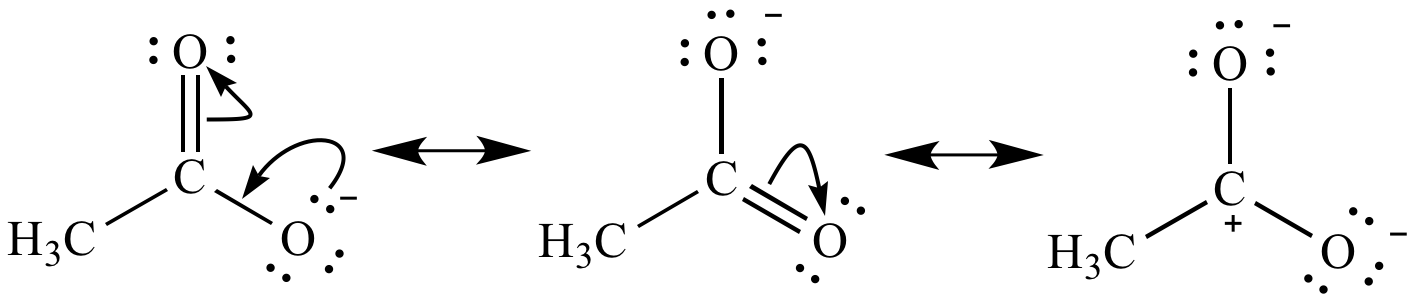
Finally, but arrows are always gonna travel from regions of high density, high electron density toe, low electron density. So that means that once I figure out my resin structures, I link them together using those double sided arrows like I have here and then brackets like I have here. We're gonna use double sided arrows and brackets toe link related structures together. Okay, um, what we're gonna do is after we've built our resident structures. What that means is that two electrons that represents two electrons are moving from one place to another. Okay, so notice that I'm using a full arrow, I'm curving it around. So what a curved arrow would look like is like this. We're gonna keep using these rules any time that we're moving electrons, which is pretty much all the time. Just so you know, these rules are gonna apply to the rest of organic can. First of all, on, we're gonna use curved arrows to represent electron movement. Okay, so let's go ahead and learn some rules. So what that means is the molecule is a blend of all the different possible resident structures that a molecule can have. And what we're gonna find out is that none of these contributing structures are actually gonna look like the actual molecules. We're gonna find out that there's something called contributing structures contributing structures or structures that both contribute to the actual representation of the molecule because they averaged together. Okay, now, something about resonant structures. And I'm also moving where lone pairs air at okay and that has to do with the electrons that are moving throughout the molecule. All in moving is double bonds around or triple bonds around. Okay, So what that means is that literally I'm not moving any atoms. Okay, the only thing that moves is the electrons, okay? And when I talk about electrons, what I'm talking about is pi Bonds pi bonds move, and I'm also talking about lone pairs. The only thing that changes is the kind of electrons that air in between them that are keeping them linked together. The reason is because remember that I said the connectivity of those atoms, how they're connected to each other doesn't change. All right, So the first thing to know is that atoms will never, ever move. So I'm gonna teach us some rules, and you guys are gonna get the hang of it as I go along. And that's what residents theory is all about. The electrons between them can move sometimes.  
So what that means is that it turns out that even though the connectivity or how atoms are connected isn't going to change. Residents theory is usedto represent the different ways that the same molecule can distribute its electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
